DeviceWatch
Automated FDA MAUDE adverse event surveillance for medical device companies. AI-powered clinical narrative analysis with compliance-ready reporting.
See It in Action
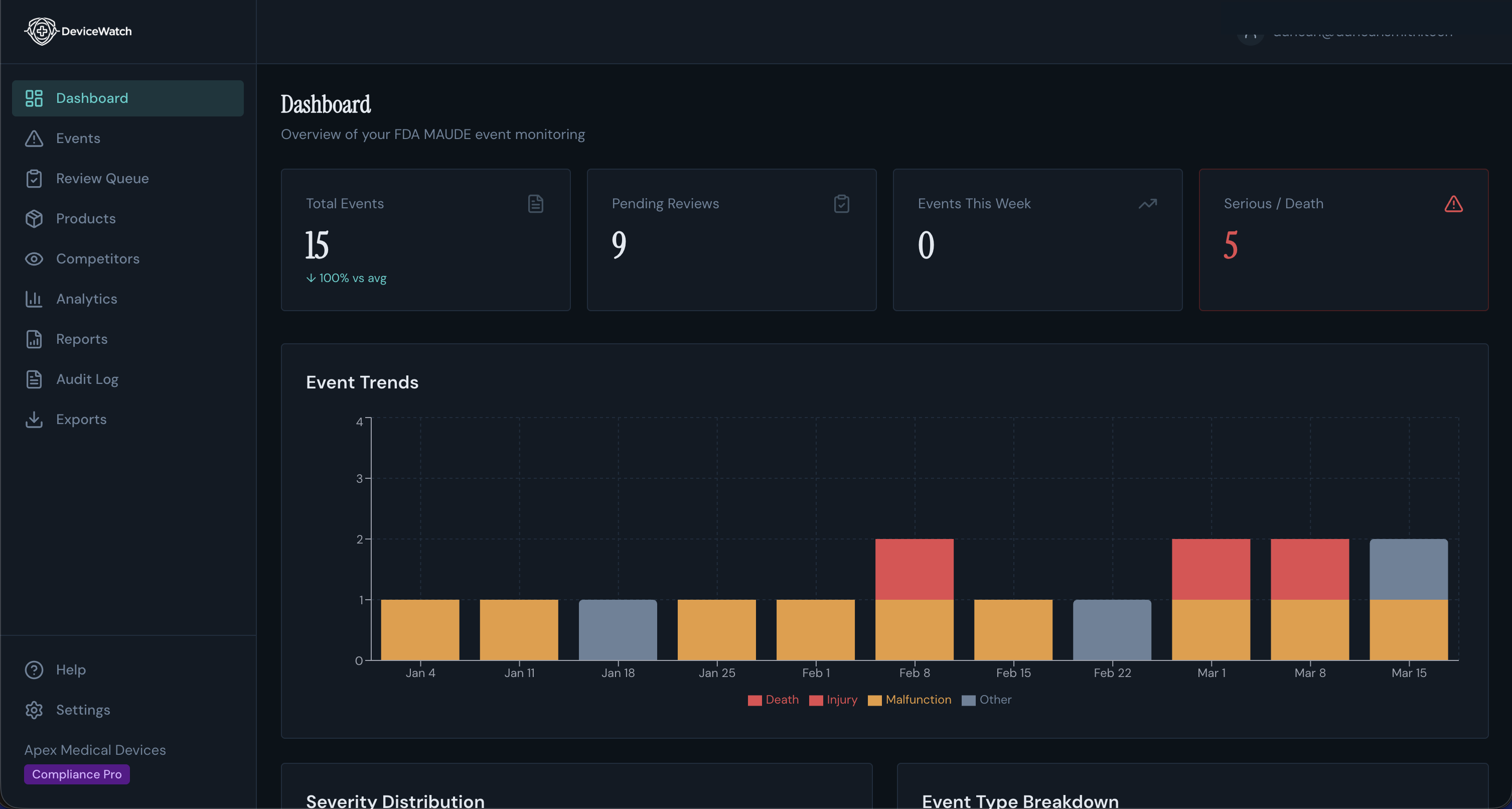
Dashboard with real-time event trends and surveillance stats
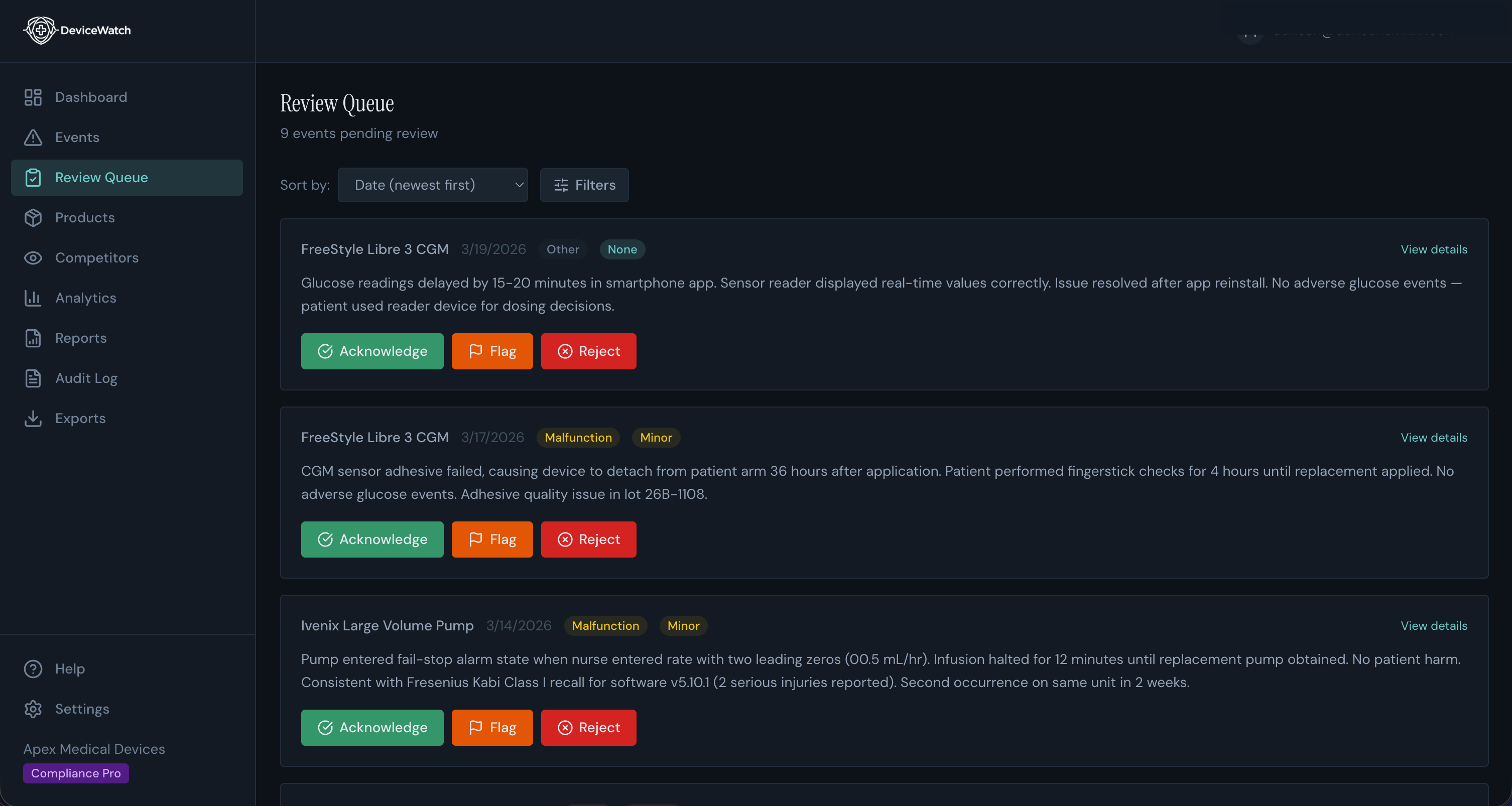
Review queue with one-click triage actions
Engineering a Regulated SaaS
The Problem
Medical device companies are required to monitor FDA MAUDE adverse event reports for their products. Most do this manually, scrolling through government databases and compiling spreadsheets. It's slow, error-prone, and doesn't scale.
The Architecture
Built on Next.js with Supabase for auth and data, DeviceWatch ingests MAUDE reports automatically, then uses Claude AI to parse clinical narratives, identify failure modes, classify severity, and generate compliance-ready reports (PSUR, Trend Analysis, CAPA).
Key Challenges
21 CFR Part 11 compliance required a full audit trail, role-based access control, and data integrity guarantees. The AI analysis pipeline needed to be deterministic enough for regulatory use while still handling the messy, unstructured language of clinical narratives.
What I Learned
Building for regulated industries means every feature decision has compliance implications. Stripe billing, Resend transactional emails, and Supabase RLS policies all had to be auditable. The AI prompts went through dozens of iterations to produce consistently structured clinical assessments.
Built with
See the Full Product
Visit the marketing site for features, pricing, and a free trial.